Logistic regression is a predictive modelling algorithm that is used when the Y variable is binary categorical. That is, it can take only two values like 1 or 0. The goal is to determine a mathematical equation that can be used to predict the probability of event 1. Once the equation is established, it can be used to predict the Y when only the Xs are known.
1. Introduction to Logistic Regression
Earlier you saw what is linear regression and how to use it to predict continuous Y variables.
In linear regression the Y variable is always a continuous variable. If suppose, the Y variable was categorical, you cannot use linear regression model it.
So what would you do when the Y is a categorical variable with 2 classes?
Logistic regression can be used to model and solve such problems, also called as binary classification problems.
A key point to note here is that Y can have 2 classes only and not more than that. If Y has more than 2 classes, it would become a multi class classification and you can no longer use the vanilla logistic regression for that.
Yet, Logistic regression is a classic predictive modelling technique and still remains a popular choice for modelling binary categorical variables.
Another advantage of logistic regression is that it computes a prediction probability score of an event. More on that when you actually start building the models.
Building the model and classifying the Y is only half work done. Actually, not even half. Because, the scope of evaluation metrics to judge the efficacy of the model is vast and requires careful judgement to choose the right model. In the next part, I will discuss various evaluation metrics that will help to understand how well the classification model performs from different perspectives.
2. Some real world examples of binary classification problems
You might wonder what kind of problems you can use logistic regression for.
Here are some examples of binary classification problems:
- Spam Detection : Predicting if an email is Spam or not
- Credit Card Fraud : Predicting if a given credit card transaction is fraud or not
- Health : Predicting if a given mass of tissue is benign or malignant
- Marketing : Predicting if a given user will buy an insurance product or not
- Banking : Predicting if a customer will default on a loan.
3. Why not linear regression?
When the response variable has only 2 possible values, it is desirable to have a model that predicts the value either as 0 or 1 or as a probability score that ranges between 0 and 1.
Linear regression does not have this capability. Because, If you use linear regression to model a binary response variable, the resulting model may not restrict the predicted Y values within 0 and 1.
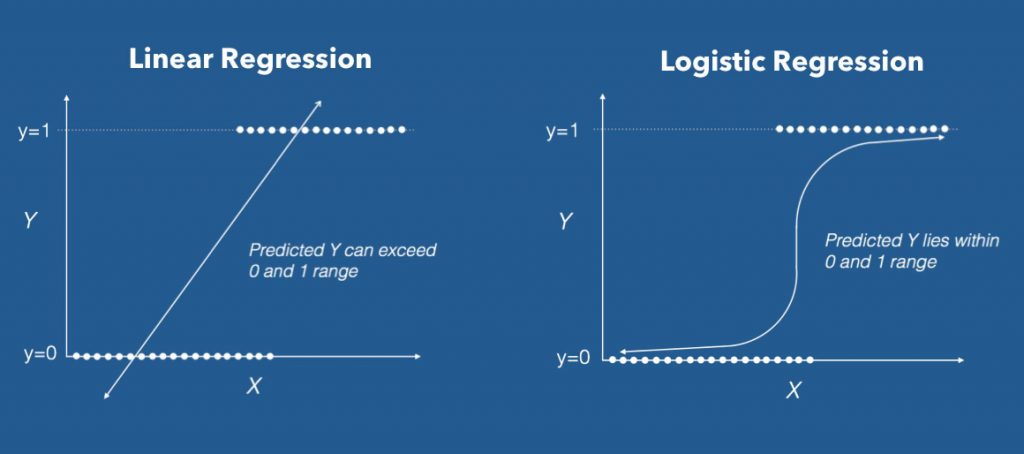
4. The Logistic Equation
Logistic regression achieves this by taking the log odds of the event ln(P/1?P), where, P is the probability of event. So P always lies between 0 and 1.
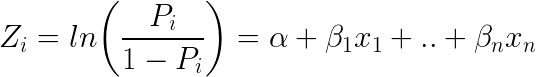
Taking exponent on both sides of the equation gives:
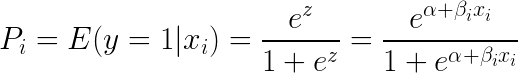
You can implement this equation using the glm() function by setting the family argument to "binomial".
# Template code
# Step 1: Build Logit Model on Training Dataset
logitMod <- glm(Y ~ X1 + X2, family="binomial", data = trainingData)
# Step 2: Predict Y on Test Dataset
predictedY <- predict(logitMod, testData, type="response") Also, an important caveat is to make sure you set the type="response" when using the predict function on a logistic regression model. Else, it will predict the log odds of P, that is the Z value, instead of the probability itself.
5. How to build logistic regression model in R?
Now let’s see how to implement logistic regression using the BreastCancer dataset in mlbench package. You will have to install the mlbench package for this.
The goal here is to model and predict if a given specimen (row in dataset) is benign or malignant, based on 9 other cell features. So, let’s load the data and keep only the complete cases.
# install.packages("mlbench")
data(BreastCancer, package="mlbench")
bc <- BreastCancer[complete.cases(BreastCancer), ] # create copy
The dataset has 699 observations and 11 columns. The Class column is the response (dependent) variable and it tells if a given tissue is malignant or benign.
Let’s check the structure of this dataset.
str(bc)
#> 'data.frame': 683 obs. of 11 variables:
#> $ Id : chr "1000025" "1002945" "1015425" "1016277" ...
#> $ Cl.thickness : Ord.factor w/ 10 levels "1"<"2"<"3"<"4"<..: 5 5 3 6 4 8 1 2 2 4 ...
#> $ Cell.size : Ord.factor w/ 10 levels "1"<"2"<"3"<"4"<..: 1 4 1 8 1 10 1 1 1 2 ...
#> $ Cell.shape : Ord.factor w/ 10 levels "1"<"2"<"3"<"4"<..: 1 4 1 8 1 10 1 2 1 1 ...
#> $ Marg.adhesion : Ord.factor w/ 10 levels "1"<"2"<"3"<"4"<..: 1 5 1 1 3 8 1 1 1 1 ...
#> $ Epith.c.size : Ord.factor w/ 10 levels "1"<"2"<"3"<"4"<..: 2 7 2 3 2 7 2 2 2 2 ...
#> $ Bare.nuclei : Factor w/ 10 levels "1","2","3","4",..: 1 10 2 4 1 10 10 1 1 1 ...
#> $ Bl.cromatin : Factor w/ 10 levels "1","2","3","4",..: 3 3 3 3 3 9 3 3 1 2 ...
#> $ Normal.nucleoli: Factor w/ 10 levels "1","2","3","4",..: 1 2 1 7 1 7 1 1 1 1 ...
#> $ Mitoses : Factor w/ 9 levels "1","2","3","4",..: 1 1 1 1 1 1 1 1 5 1 ...
#> $ Class : Factor w/ 2 levels "benign","malignant": 1 1 1 1 1 2 1 1 1 1 ...Except Id, all the other columns are factors. This is a problem when you model this type of data.
Because, when you build a logistic model with factor variables as features, it converts each level in the factor into a dummy binary variable of 1’s and 0’s.
For example, Cell shape is a factor with 10 levels. When you use glm to model Class as a function of cell shape, the cell shape will be split into 9 different binary categorical variables before building the model.
If you are to build a logistic model without doing any preparatory steps then the following is what you might do. But we are not going to follow this as there are certain things to take care of before building the logit model.
The syntax to build a logit model is very similar to the lm function you saw in linear regression. You only need to set the family='binomial' for glm to build a logistic regression model.
glm stands for generalised linear models and it is capable of building many types of regression models besides linear and logistic regression.
Lets see how the code to build a logistic model might look like. I will be coming to this step again later as there are some preprocessing steps to be done before building the model.
glm(Class ~ Cell.shape, family="binomial", data = bc)
#> Call: glm(formula = Class ~ Cell.shape, family = "binomial", data = bc)
#> Coefficients:
#> (Intercept) Cell.shape.L Cell.shape.Q Cell.shape.C
#> 4.189 20.911 6.848 5.763
#> Cell.shape^4 Cell.shape^5 Cell.shape^6 Cell.shape^7
#> -1.267 -4.439 -5.183 -3.013
#> Cell.shape^8 Cell.shape^9
#> -1.289 -0.860
#> Degrees of Freedom: 682 Total (i.e. Null); 673 Residual
#> Null Deviance: 884.4
#> Residual Deviance: 243.6 AIC: 263.6In above model, Class is modeled as a function of Cell.shape alone.
But note from the output, the Cell.Shape got split into 9 different variables. This is because, since Cell.Shape is stored as a factor variable, glm creates 1 binary variable (a.k.a dummy variable) for each of the 10 categorical level of Cell.Shape.
Clearly, from the meaning of Cell.Shape there seems to be some sort of ordering within the categorical levels of Cell.Shape. That is, a cell shape value of 2 is greater than cell shape 1 and so on.
This is the case with other variables in the dataset a well. So, its preferable to convert them into numeric variables and remove the id column.
Had it been a pure categorical variable with no internal ordering, like, say the sex of the patient, you may leave that variable as a factor itself.
# remove id column
bc <- bc[,-1]
# convert factors to numeric
for(i in 1:9) {
bc[, i] <- as.numeric(as.character(bc[, i]))
}Another important point to note. When converting a factor to a numeric variable, you should always convert it to character and then to numeric, else, the values can get screwed up.
Now all the columns are numeric.
Also I’d like to encode the response variable into a factor variable of 1’s and 0’s. Though, this is only an optional step.
So whenever the Class is malignant, it will be 1 else it will be 0. Then, I am converting it into a factor.
bc$Class <- ifelse(bc$Class == "malignant", 1, 0)
bc$Class <- factor(bc$Class, levels = c(0, 1))The response variable Class is now a factor variable and all other columns are numeric.
Alright, the classes of all the columns are set. Let’s proceed to the next step.
6. How to deal with Class Imbalance?
Before building the logistic regressor, you need to randomly split the data into training and test samples.
Since the response variable is a binary categorical variable, you need to make sure the training data has approximately equal proportion of classes.
table(bc$Class)
#> benign malignant
#> 444 239The classes ‘benign’ and ‘malignant’ are split approximately in 1:2 ratio.
Clearly there is a class imbalance. So, before building the logit model, you need to build the samples such that both the 1’s and 0’s are in approximately equal proportions.
This concern is normally handled with a couple of techniques called:
- Down Sampling
- Up Sampling
- Hybrid Sampling using SMOTE and ROSE.
So, what is Down Sampling and Up Sampling?
7. How to handle Class Imbalance with Upsampling and Downsampling
In Down sampling, the majority class is randomly down sampled to be of the same size as the smaller class. That means, when creating the training dataset, the rows with the benign Class will be picked fewer times during the random sampling.
Similarly, in UpSampling, rows from the minority class, that is, malignant is repeatedly sampled over and over till it reaches the same size as the majority class (benign).
But in case of Hybrid sampling, artificial data points are generated and are systematically added around the minority class. This can be implemented using the SMOTE and ROSE packages.
However for this example, I will show how to do up and down sampling.
So let me create the Training and Test Data using caret Package.
library(caret)
'%ni%' <- Negate('%in%') # define 'not in' func
options(scipen=999) # prevents printing scientific notations.
# Prep Training and Test data.
set.seed(100)
trainDataIndex <- createDataPartition(bc$Class, p=0.7, list = F) # 70% training data
trainData <- bc[trainDataIndex, ]
testData <- bc[-trainDataIndex, ]In the above snippet, I have loaded the caret package and used the createDataPartition function to generate the row numbers for the training dataset. By setting p=.70I have chosen 70% of the rows to go inside trainData and the remaining 30% to go to testData.
table(trainData$Class)There is approximately 2 times more benign samples. So lets downsample it using the downSample function from caret package.
To do this you just need to provide the X and Y variables as arguments.
# Down Sample
set.seed(100)
down_train <- downSample(x = trainData[, colnames(trainData) %ni% "Class"],
y = trainData$Class)
table(down_train$Class)
#> benign malignant
#> 168 168Benign and malignant are now in the same ratio.
The %ni% is the negation of the %in% function and I have used it here to select all the columns except the Class column.
The downSample function requires the ‘y’ as a factor variable, that is reason why I had converted the class to a factor in the original data.
Great! Now let me do the upsampling using the upSample function. It follows a similar syntax as downSample.
# Up Sample.
set.seed(100)
up_train <- upSample(x = trainData[, colnames(trainData) %ni% "Class"],
y = trainData$Class)
table(up_train$Class)
#> benign malignant
#> 311 311As expected, benign and malignant are now in the same ratio.
I will use the downSampled version of the dataset to build the logit model in the next step.
8. Building the Logistic Regression Model
# Build Logistic Model
logitmod <- glm(Class ~ Cl.thickness + Cell.size + Cell.shape, family = "binomial", data=down_train)
summary(logitmod)
#> Call:
#> glm(formula = Class ~ Cl.thickness + Cell.size + Cell.shape,
#> family = "binomial", data = down_train)
#>
#> Deviance Residuals:
#> Min 1Q Median 3Q Max
#> -2.1136 -0.0781 -0.0116 0.0000 3.9883
#> Coefficients:
#> Estimate Std. Error z value Pr(>|z|)
#> (Intercept) 21.942 3949.431 0.006 0.996
#> Cl.thickness.L 24.279 5428.207 0.004 0.996
#> Cl.thickness.Q 14.068 3609.486 0.004 0.997
#> Cl.thickness.C 5.551 3133.912 0.002 0.999
#> Cl.thickness^4 -2.409 5323.267 0.000 1.000
#> Cl.thickness^5 -4.647 6183.074 -0.001 0.999
#> Cl.thickness^6 -8.684 5221.229 -0.002 0.999
#> Cl.thickness^7 -7.059 3342.140 -0.002 0.998
#> Cl.thickness^8 -2.295 1586.973 -0.001 0.999
#> Cl.thickness^9 -2.356 494.442 -0.005 0.996
#> Cell.size.L 28.330 9300.873 0.003 0.998
#> Cell.size.Q -9.921 6943.858 -0.001 0.999
#> Cell.size.C -6.925 6697.755 -0.001 0.999
#> Cell.size^4 6.348 10195.229 0.001 1.000
#> Cell.size^5 5.373 12153.788 0.000 1.000
#> Cell.size^6 -3.636 10824.940 0.000 1.000
#> Cell.size^7 1.531 8825.361 0.000 1.000
#> Cell.size^8 7.101 8508.873 0.001 0.999
#> Cell.size^9 -1.820 8537.029 0.000 1.000
#> Cell.shape.L 10.884 9826.816 0.001 0.999
#> Cell.shape.Q -4.424 6049.000 -0.001 0.999
#> Cell.shape.C 5.197 6462.608 0.001 0.999
#> Cell.shape^4 12.961 10633.171 0.001 0.999
#> Cell.shape^5 6.114 12095.497 0.001 1.000
#> Cell.shape^6 2.716 11182.902 0.000 1.000
#> Cell.shape^7 3.586 8973.424 0.000 1.000
#> Cell.shape^8 -2.459 6662.174 0.000 1.000
#> Cell.shape^9 -17.783 5811.352 -0.003 0.998
#> (Dispersion parameter for binomial family taken to be 1)
#> Null deviance: 465.795 on 335 degrees of freedom
#> Residual deviance: 45.952 on 308 degrees of freedom
#> AIC: 101.95
#> Number of Fisher Scoring iterations: 219. How to Predict on Test Dataset
The logitmod is now built. You can now use it to predict the response on testData.
pred <- predict(logitmod, newdata = testData, type = "response")Now, pred contains the probability that the observation is malignant for each observation.
Note that, when you use logistic regression, you need to set type='response' in order to compute the prediction probabilities. This argument is not needed in case of linear regression.
The common practice is to take the probability cutoff as 0.5. If the probability of Y is > 0.5, then it can be classified an event (malignant).
So if pred is greater than 0.5, it is malignant else it is benign.
y_pred_num <- ifelse(pred > 0.5, 1, 0)
y_pred <- factor(y_pred_num, levels=c(0, 1))
y_act <- testData$ClassLet’s compute the accuracy, which is nothing but the proportion of y_pred that matches with y_act.
mean(y_pred == y_act) # 94+%
#> [1] 0.9411765There you have an accuracy rate of 94%.
10. Why handling with class imbalance is important?
Alright I promised I will tell you why you need to take care of class imbalance earlier. To understand that lets assume you have a dataset where 95% of the Y values belong to benign class and 5% belong to malignant class.
Had I just blindly predicted all the data points as benign, I would achieve an accuracy percentage of 95%. Which sounds pretty high. But obviously that is flawed. What matters is how well you predict the malignant classes.
So that requires the benign and malignant classes are balanced AND on top of that I need more refined accuracy measures and model evaluation metrics to improve my prediction model.
Full Code
# Load data
# install.packages('mlbench')
data(BreastCancer, package="mlbench")
bc <- BreastCancer[complete.cases(BreastCancer), ] # keep complete rows
# remove id column
bc <- bc[,-1]
# convert to numeric
for(i in 1:9) {
bc[, i] <- as.numeric(as.character(bc[, i]))
}
# Change Y values to 1's and 0's
bc$Class <- ifelse(bc$Class == "malignant", 1, 0)
bc$Class <- factor(bc$Class, levels = c(0, 1))
# Prep Training and Test data.
library(caret)
'%ni%' <- Negate('%in%') # define 'not in' func
options(scipen=999) # prevents printing scientific notations.
set.seed(100)
trainDataIndex <- createDataPartition(bc$Class, p=0.7, list = F)
trainData <- bc[trainDataIndex, ]
testData <- bc[-trainDataIndex, ]
# Class distribution of train data
table(trainData$Class)
# Down Sample
set.seed(100)
down_train <- downSample(x = trainData[, colnames(trainData) %ni% "Class"],
y = trainData$Class)
table(down_train$Class)
# Up Sample (optional)
set.seed(100)
up_train <- upSample(x = trainData[, colnames(trainData) %ni% "Class"],
y = trainData$Class)
table(up_train$Class)
# Build Logistic Model
logitmod <- glm(Class ~ Cl.thickness + Cell.size + Cell.shape, family = "binomial", data=down_train)
summary(logitmod)
pred <- predict(logitmod, newdata = testData, type = "response")
pred
# Recode factors
y_pred_num <- ifelse(pred > 0.5, 1, 0)
y_pred <- factor(y_pred_num, levels=c(0, 1))
y_act <- testData$Class
# Accuracy
mean(y_pred == y_act) # 94%11. Conclusion
In this post you saw when and how to use logistic regression to classify binary response variables in R.
You saw this with an example based on the BreastCancer dataset where the goal was to determine if a given mass of tissue is malignant or benign.
Building the model and classifying the Y is only half work done. Actually, not even half. Because, the scope of evaluation metrics to judge the efficacy of the model is vast and requires careful judgement to choose the right model. In the next part, I will discuss various evaluation metrics that will help to understand how well the classification model performs from different perspectives.






